These thermometers are used for temperature measurements from -200 to 750 °C. They are contact-type thermometers. Fig. 1 shows the principle of their design.
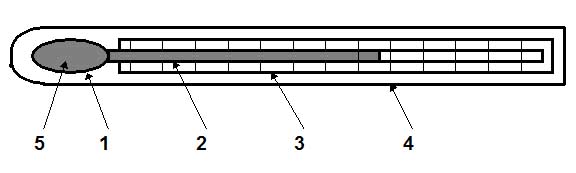
Figure 1.: Liquid-in-glass thermometer
This thermometer consists of a glass bulb 1, which is connected with a glass capillary tube 2. A scale 3 in degrees of Celsius or Fahrenheit is placed behind the capillary tube. The bulb, the capillary tube and the scale are placed in a glass tube 4 to protect them against the damage.
A thermometric liquid 5 fills the bulb and a part of the capillary tube. The operational principle of these thermometers is based on the difference between the volume expansion of liquids and glass with temperature. The relationship that governs the operation of this device is
![]()
where,
VT - volume of liquid at temperature T, m3;
VT - volume of liquid at temperature T0, m3;
∆T= T-T0 - difference of temperatures, K;
ᵦ - volumetric thermal expansion coefficient, 1/K.
The volumetric thermal expansion coefficient of glass is much less than that of liquids. The variation of temperature (up and down) of the bulb causes liquid in the system to expand or decrease its volume, respectively.
As a result of such changes (the internal volume of the glass bulb and the glass capillary varies negligible), the length of the liquid column in the capillary tube goes up or down proportionally to the variation of temperature.
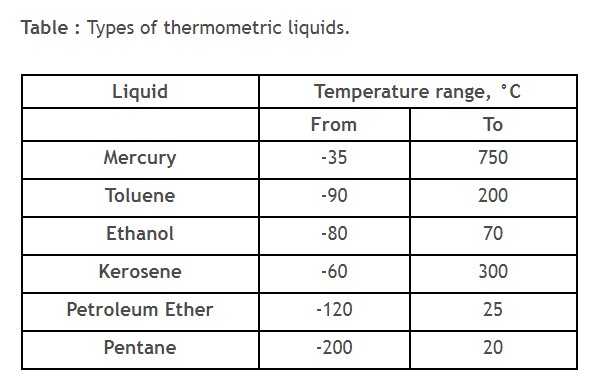
The type of thermometric liquid depends on the lower and upper limits of the measuring temperature range.
Types of thermometric liquids
Among these liquids mercury is the most widely used, because:
• mercury is easy obtainable with high chemical purity;
• mercury does not wet glass (this increases the accuracy of measurement/ reading);
• mercury remains in liquid state in a wide temperature range.
Among disadvantages inherent to mercury-in-glass thermometers we can mention the following:
• mercury is a poisonous element, which affects the central and peripheral nervous system, its vapour is the most toxic;
• small volumetric thermal expansion coefficient for mercury, therefore, mercury is used in thermometers with capillaries of small internal diameter;
The solidifying point of mercury, ie 38 °C, limits the lowest temperature that can be measured by mercury-in-glass thermometers. The upper temperature is determined by the temperature at which glass still retains its solid properties. This temperature is equal about 600 °C for glass, and about 750 °C for silicon glass.
When air above mercury in the capillary is removed, a mercury-in-glass thermometer can be used at temperatures below 300 °C, because the boiling temperature of mercury at atmospheric pressure is equal 356.9 °C.
In order to increase this temperature range it is necessary to increase the boiling temperature of mercury (saturation temperature). This can be achieved by increasing pressure in the capillary. Usually, the space above mercury in the capillary is filled by inert gas (such as nitrogen, argon) under pressure.
Liquid-in-glass thermometers with organic thermometric liquids are used for temperature measurements from -200 to 200 °C.
One advantage of these thermometers is:
• a higher volume thermal expansion coefficient comparing with that for mercury (six times higher in average).
Disadvantage of thermometers with organic liquids is:
• these liquids wet glass, therefore, in order to increase the accuracy of measurement/reading, glass capillaries with bigger internal diameters (up to 1 mm) are used.
Advantages of liquid-in-glass thermometers are as follows:
• they are simple in design;
• they are relatively highly accurate in temperature measurement.
There are several disadvantages inherent to liquid-in-glass thermometers
• they are fragile;
• it is difficult to perform readings due to low visibility of the scale;
• they are not capable of distance transmission of a measuring signal, therefore, they are used as locally placed devices;
• impossibility to repair;
• high values of time lag;
• low visibility of mercury in the capillary.