Using nitrogen safely
Nitrogen is often mistakenly considered harmless because it is non-toxic and largely inert.
However, nitrogen can act as an asphyxiant by displacing oxygen in air to levels below those required for survival.
Therefore, use caution when handling nitrogen. Reference 5 provides more information about the safe handling of nitrogen.
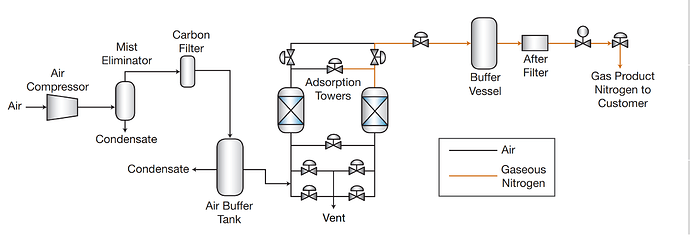
Nitrogen-PSA Plant
Typical applications
Because nitrogen generated by PSA generally contains small amounts of oxygen, some types of chemical processes lend themselves better than others to this type of supply.
Manufacturing pressure-sensitive adhesives.
The manufacture of pressure-sensitive adhesives involves flammable solvents and powders that present some safety hazards. In this process, finely ground rubber and fillers are added to flammable organic solvents in agitated tanks.
Because vapors from the solvents are typically flammable in air, and the powders can generate significant sparks from the buildup of static electricity, reactions involving these materials can be dangerous in the presence of air (i.e., 20.9% oxygen). For these reasons, an adhesives manufacturer was compelled to take steps to reduce the risk of explosions and fires.
First, the company installed static electricity eliminators. However, as these devices do not provide complete protection, occasional sparking was
still a risk.
Consequently, the manufacturer installed a nitrogen purge system to reduce the oxygen content in the tanks to a nonflammable level of no more than 2.5%. A PSA system that produced nitrogen containing 2% oxygen was combined with an inerting system to achieve a
safe operating atmosphere.
Storing and handling free-radical polymerization monomers.
In some situations, it is desirable to have small
amounts of oxygen present in an inert nitrogen atmosphere. For these applications, if the nitrogen is delivered as a liquid and then vaporized, some amount of air must be blended with the high-purity nitrogen to achieve the desired oxygen concentration. However, PSA nitrogen generators are a more economical and convenient alternative, as they provide nitrogen with a small oxygen content.
One manufacturer’s process involved the storage and handling of monomers prior to free-radical polymerization. Monomer vapors are highly flammable and require an inert atmosphere in the storage headspace to prevent a safety hazard. The monomers contained hydroquinone as an inhibitor to prevent premature polymerization during storage.
To keep the hydroquinone active and to ensure the safe handling of the materials, a small volume of oxygen was required in the inert nitrogen gas. Two options could provide an inerting atmosphere with 2–4% oxygen: use liquid nitrogen, continuously blending it with oxygen, or install a PSA unit to provide a nitrogen product with the necessary oxygen level. The manufacturer selected a PSA system, which produced the required atmosphere and eliminated the additional blending steps.
Handling vinyl chloride monomer.
Vinyl chloride monomer (VCM) is an important industrial chemical because of its use in a variety of products, including low-cost polymers. Almost all of the vinyl chloride produced goes into the
manufacture of polyvinyl chloride (PVC), which is used in automotive parts, packaging, pipes, construction materials, furniture, and numerous other products. VCM is extremely flammable as both a vapor and a liquid. When VCM is stored in closed containers, a flammable atmosphere can develop.
Flammable mixtures of VCM can
be readily ignited, even by static discharge (7).
The LFL for VCM is 3.8% (7). T
o prevent flammability, 25% of the LFL, which corresponds to 0.95% VCM or 99.05% nitrogen by volume, may be considered a sufficient atmosphere. However, because VCM can undergo polymerization or decomposition under certain conditions and in the presence of moisture and oxygen, a higher-purity nitrogen atmosphere was recommended.
Because of the application’s purity and pattern requirements, a VCM manufacturer installed an inerting system with nitrogen supplied by a PSA unit, which provides 99.5% nitrogen.